The general secretary for Bradford Hindu Council is taking part in the world’s first antiviral Covid-19 study by UK Government.
Adults over the age of 50 or with an underlying health condition who test positive for Covid-19 are being urged to sign up for a world-first antiviral Covid-19 study which is providing life-saving antivirals to thousands of people.
The Government and leading charities, including Kidney Care UK, Cystic Fibrosis Trust, Diabetes UK and the British Liver Trust, are calling on at least 6,000 more participants to come forward for these cutting-edge treatments through the Panoramic study.

This is so that expert scientists can understand more about how to deploy these treatments in the NHS more widely later in the year – including who would benefit most from receiving antiviral treatments for Covid-19.
Antivirals are medicines that can be swallowed as a tablet to help treat people with Covid-19 infections to reduce the risk of hospitalisations and death.
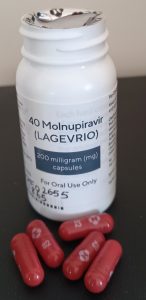
Molnupiravir, which is currently being deployed through the study, has shown to reduce this for at-risk, non-hospitalised adults with mild to moderate Covid-19 by 30% – potentially saving thousands of lives once the drugs are available to the NHS.
Harkishan Mistry, the general secretary for Bradford Hindu Council 58, tested positive for Covid-19 last Monday. After a quick phone call, he was selected for the trial and is now taking four anti-viral capsules in the morning and four capsules in the evening, 12 hours apart for five days.
He has to fill a two-minute survey out every day for 28 days to rate his symptoms and at the end of the trial will receive a gift card in thanks for his participation.
Mr Mistry said: “After testing positive for COVID-19 last Monday on a lateral flow, a friend recommended I look into the antivirals trial.
“I spoke to a woman for twenty minutes on the phone or took all my details and explained the trial details.
“As I’m over 50 and was experiencing symptoms such as sore throat, headache and achiness, I was eligible and was selected to receive the antiviral treatment by Monday afternoon.
“The whole process was so simple – a courier delivered the capsules the next morning and I began taking the course straight away. While I’m still testing positive and remain in isolation, my symptoms eased daily and I feel much better.
“I’d really recommend anyone who’s eligible for the trial to sign up – why would you not if we can help others and ease pressure on the NHS.”
Health and Social Care Secretary Sajid Javid said: “The vaccines are critical as the first line of defence, but antivirals form a vital part of our approach as we learn to live with COVID by preventing the most vulnerable from being hospitalised.
“If you’re eligible, please step forward for the PANORAMIC trial and play your part in a vital mission – helping us to learn more about medicines that could save thousands of lives.”
Anyone over the age of 50 or between 18 to 49 with an underlying health condition can sign up for the study as soon as they receive a positive PCR or lateral flow test result.
They need to be experiencing COVID-19 symptoms that began in the last 5 days to be eligible to enrol.




